|
The resulting nucleus is a different nuclide from the initial nuclei. This process also releases a substantial amount of energy. Similarly, in nuclear fusion, two light nuclei combine to form a heavier nucleus. Each of the resulting nuclei is a new nuclide. For example, in nuclear fission, a heavy nucleus splits into two smaller nuclei, releasing a significant amount of energy. Reacting atomic nuclei are the initial nuclei that participate in the reaction, while produced nuclides are the new nuclei formed as a result of the reaction.ĭuring a nuclear reaction, the number of protons and neutrons in the nucleus may change, leading to the formation of different nuclides. To understand nuclear reactions, it is essential to differentiate between the reacting atomic nuclei and the resulting nuclides. Distinction between Reacting Atomic Nuclei and Produced Nuclides These processes involve the interaction of subatomic particles, such as protons, neutrons, and electrons, within the nucleus. Nuclear reactions can occur through different mechanisms, such as nuclear fission, nuclear fusion, and radioactive decay. Unlike chemical reactions that involve the rearrangement of electrons and the formation of new molecules, nuclear reactions primarily focus on changes within the nucleus itself. In simple terms, a nuclear reaction refers to any process that alters the structure or composition of atomic nuclei. Understanding nuclear reactions is crucial in various fields, including energy production, medicine, and scientific research. These reactions involve changes in the composition of atomic nuclei, resulting in the formation of different nuclides. 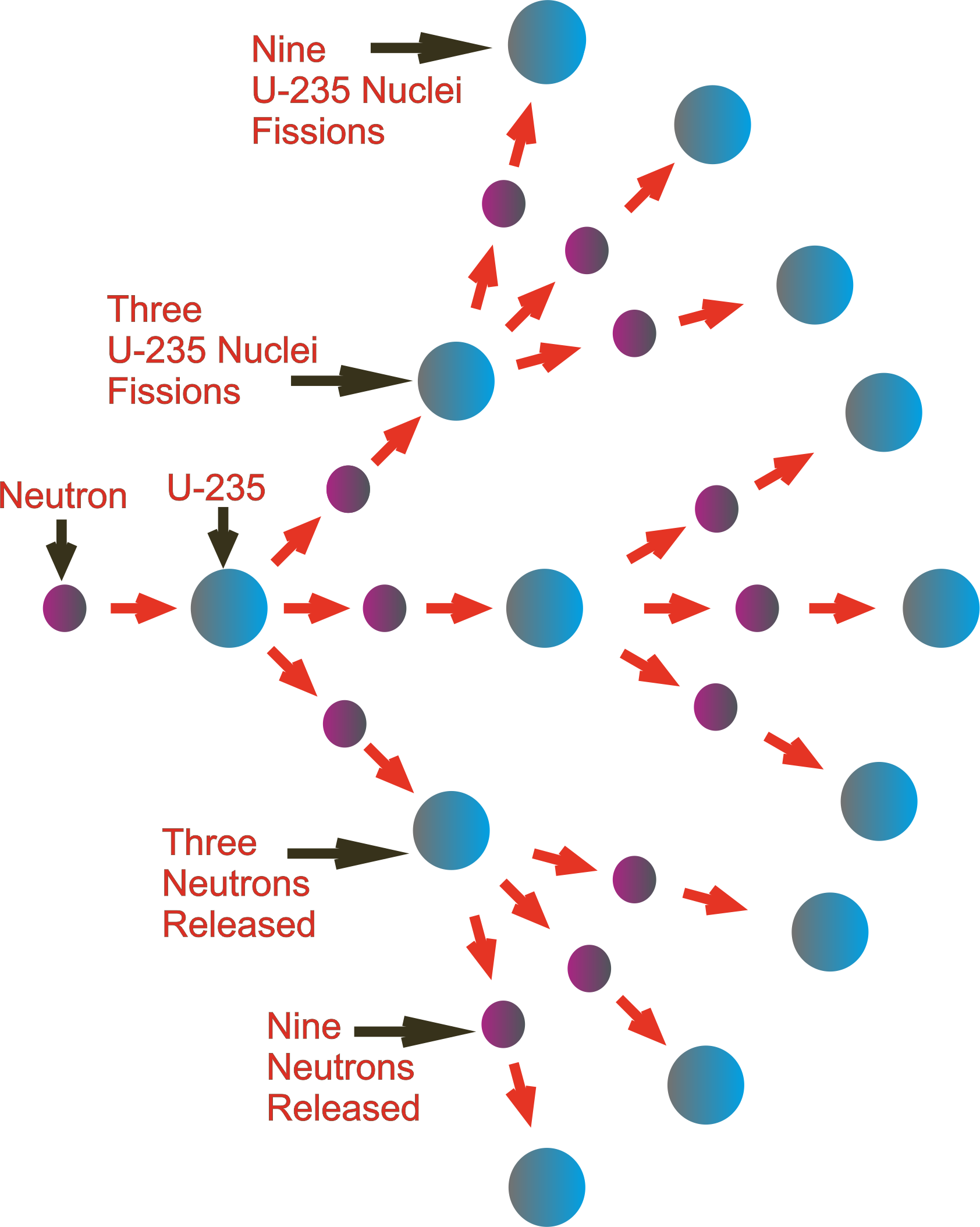
Nuclear reactions are fundamental processes that occur within the nucleus of an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
